2012国际卒中大会:新型免提超声装置与tPA联合溶栓安全有效
2012-02-17 MedSci MedSci原创
2012国际卒中大会公布的最新研究结果表明,应用新型免提超声溶栓装置与组织型纤溶酶原激活物(tPA)联用具有较好的安全性,并且有较高的血管再通率。此种联合疗法值得在Ⅲ期有效性试验中进一步评估。 当与静脉(IV)-tPA联用时,经FDA批准的手持式经颅多普勒(TCD)超声装置可改善再通。免提经颅超声和全身t-PA脑出血联合溶栓(CLOTBUST-HF)研究是首次在人类中实
2012国际卒中大会公布的最新研究结果表明,应用新型免提超声溶栓装置与组织型纤溶酶原激活物(tPA)联用具有较好的安全性,并且有较高的血管再通率。此种联合疗法值得在Ⅲ期有效性试验中进一步评估。
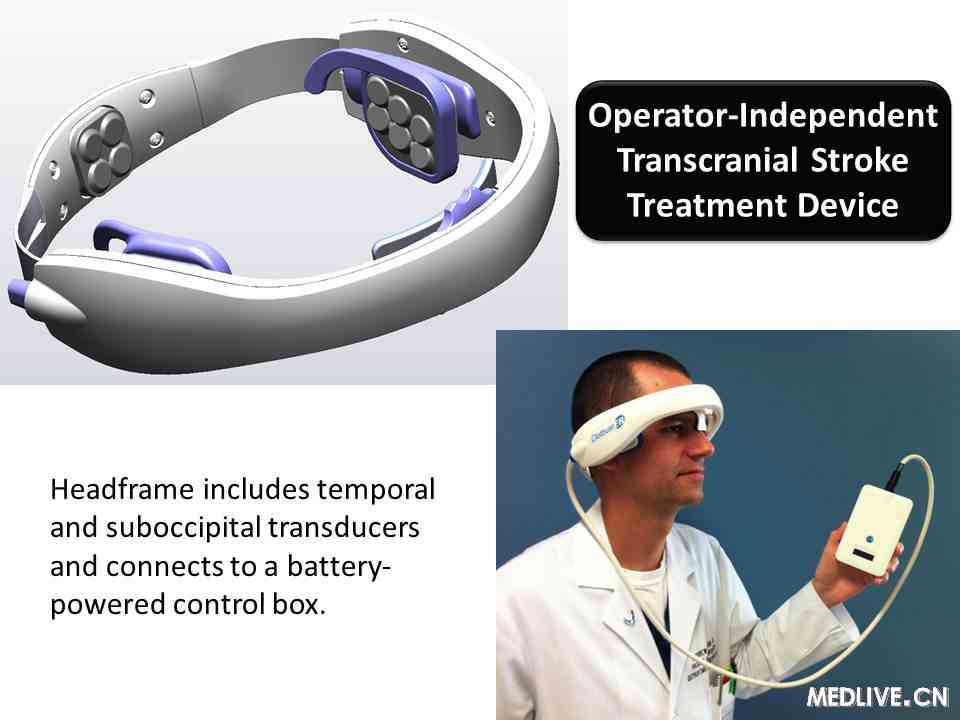
Background and Purpose: Transcranial Doppler (TCD) ultrasound utilizing hand-held, FDA-approved devices enhances recanalization when combined with IV-tPA. The Combined Lysis of Thrombus in Brain Ischemia With Transcranial Ultrasound and Systemic T-PA- Hands-Free (CLOTBUST-HF) study is a first-inman, NIH-sponsored, multi-center, open-label, pilot safety study of tPA plus a novel operator-independent ultrasound device in patients with ischemic stroke due to proximal intracranial occlusion. The hands-free device is composed of 18 ultrasound probes which are activated sequentially in order to provide therapeutic ultrasound to the entire intracranial circulation. Prior to the current study, 15 healthy volunteers tolerated the procedure without any change on MR permeability imaging.
Subjects & Methods: In the CLOTBUST-HF study (NCT#01240356), all study patients received standard dose IV tPA, and immediately after tPA bolus, 2-hours of 2-MHz therapeutic exposure to CLOTBUST-HF device was begun. The primary outcome was occurrence of symptomatic intracerebral hemorrhage (sICH). All patients underwent pre- and post-treatment assessment of vascular patency and recanalization measured by standardized TCD or CT angiography criteria. NIHSS scores were collected at 2-hours and mRS at 90 days.
Results: All pre-planned 20 patients were enrolled (60% men, mean age 63±14 years, median NIHSS = 15). Sites of pre-treatment arterial occlusion were: 14/20 (70%) MCA, 3/20 (15%) terminal ICA and 3/20 (15%) vertebral. The median (IQR) time tPA to beginning of sonothrombolysis was 22 (13.5, 29) minutes. All patients tolerated the entire 2 hours of insonation and none developed sICH. To date, no serious adverse events have been adjudicated to be related to the study device. Rates of 2 hour recanalization were: 8/20 (40%; 95% CI: 19-64) complete and 2/20 (10%; 95% CI: 1-32) partial. MCA occlusions demonstrated the greatest complete recanalization rate: 8/14 (57%; 95% CI: 29-82). 90 day mRS and NIHSS score data are pending and will be presented at the meeting. Our rate of 2-hour sustained complete recanalization closely parallels the results of the original CLOTBUST study (38%) which utilized operator-dependent hand-held technology.
Conclusion: Sonothrombolysis using a novel, operator-independent device in combination with systemic tPA appears safe and high recanalization rates warrant continued evaluation of this treatment combination in a phase III efficacy trial (CLOTBUSTER, NCT#01098981).
作者:MedSci
版权声明:
本网站所有注明“来源:梅斯医学”或“来源:MedSci原创”的文字、图片和音视频资料,版权均属于梅斯医学所有。非经授权,任何媒体、网站或个人不得转载,授权转载时须注明“来源:梅斯医学”。其它来源的文章系转载文章,本网所有转载文章系出于传递更多信息之目的,转载内容不代表本站立场。不希望被转载的媒体或个人可与我们联系,我们将立即进行删除处理。
在此留言





#tPA#
98